Scheme 2. Preparation of alkene 14 (DIPEA = N,N-diisopropylethylamine). | Download Scientific Diagram

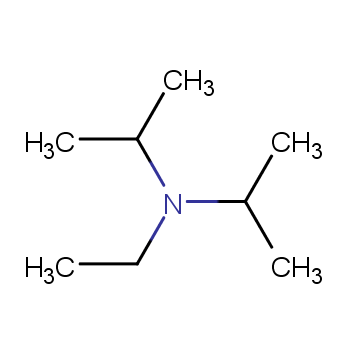
DIPEA (N,N-diisopropylethylamine, Hunig's Base) Molecule. Skeletal Formula. Royalty Free SVG, Cliparts, Vectors, And Stock Illustration. Image 149287710.

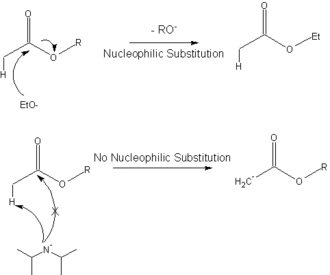
SOLVED: Predict the minor and major product DIPEA-diisopropylethylamina or Hunig s base DcJe malc prcducl rnE prcducl Predict products and indicate which one major product 4meducl ShowIne machansm oline lollowing reaclions Clearly

Electrochemical Formation of Cinnamaldehyde by the Electrolyte System N,N‐ Diisopropylethylamine and 1,1,1,3,3,3‐Hexafluoropropan‐2‐ol - Imada - 2020 - ChemElectroChem - Wiley Online Library
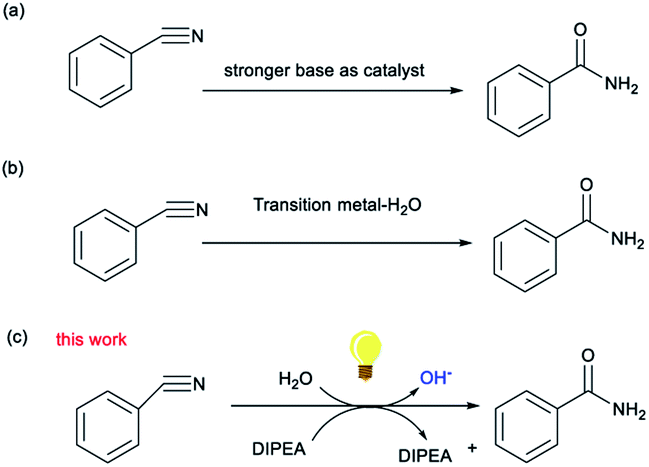
DIPEA-induced activation of OH − for the synthesis of amides via photocatalysis - RSC Advances (RSC Publishing) DOI:10.1039/D2RA02107B

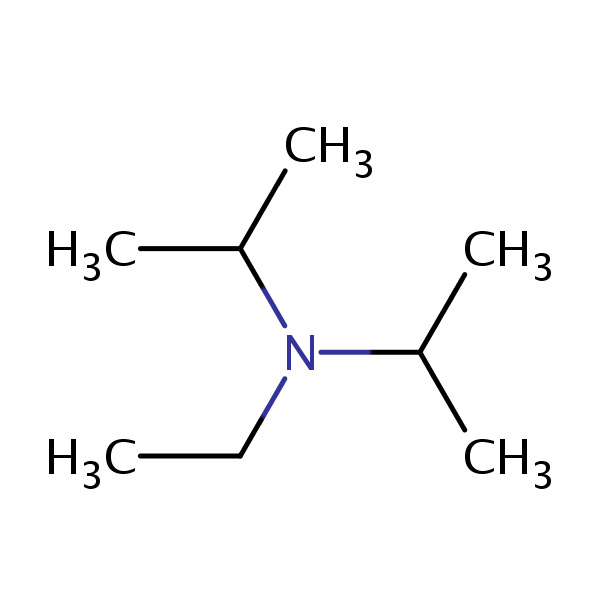
Dipea Nndiisopropylethylamine Hunigs Base Molecule Skeletal Stock Vector (Royalty Free) 1093026992 | Shutterstock

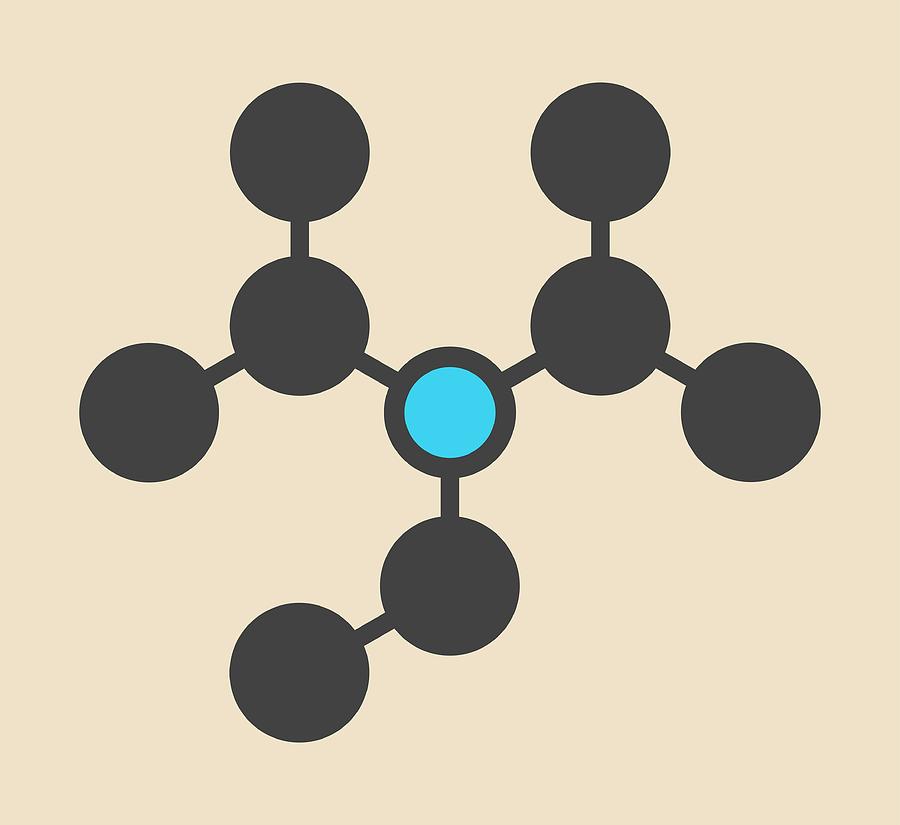
DIPEA (N,N-diisopropylethylamine, Hunig's base) molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (hidden), carbon (grey Stock Photo - Alamy

Diisopropylethylamine-triggered, highly efficient, self-catalyzed regioselective acylation of carbohydrates and diols - ScienceDirect