
SOLVED: 8. Calculate the pH of a 1.00 L aqueous solution prepared by mixing 12.43 grams of TRIS (molar mass 121.135) and 5.67 grams of TRIS hydrochloride (molar mass 157.596) (pKb of TRIS = 5.928). (6 points)

SOLVED: 1. Describe how you would make the following two buffers (remember you need to calculate the mass of Tris base and the mass of Tris-acetate needed). Please show your work. a.
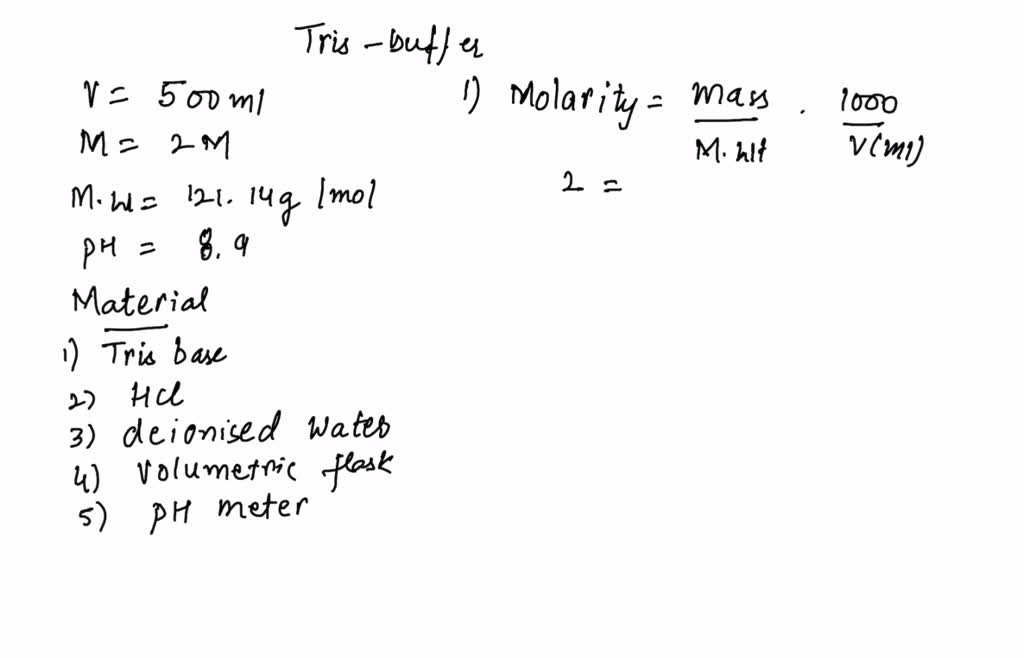


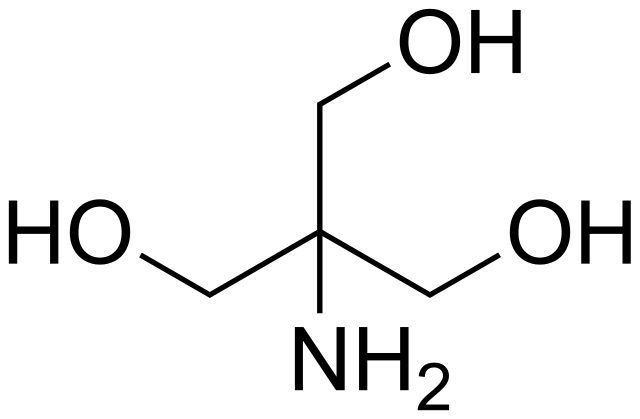






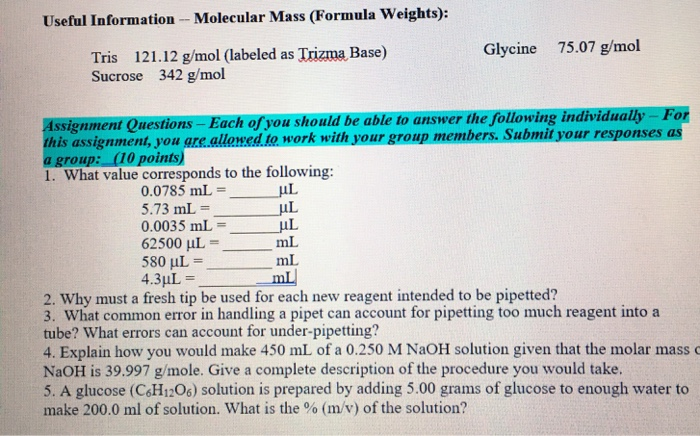



![Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/tris-base-molecular-weight-calculation-300x204.jpg)

